FuelIONIZER® Heaters
Resulting in...
- pollution* (*multiple nasty chemicals)
- excess carbon
- excess CO2
- raw fuel
all being released into the environment
It’s the Physics
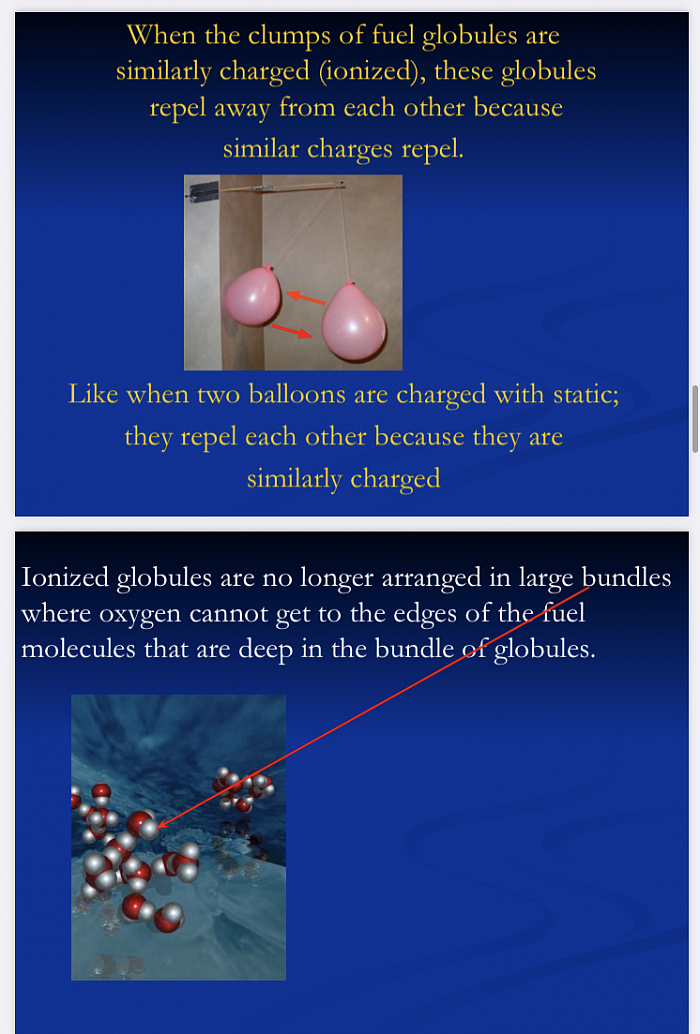
Fuels exist in “globules”
(bundles / clumps / balls of fuel molecules)-
NOT as individual fuel molecules.
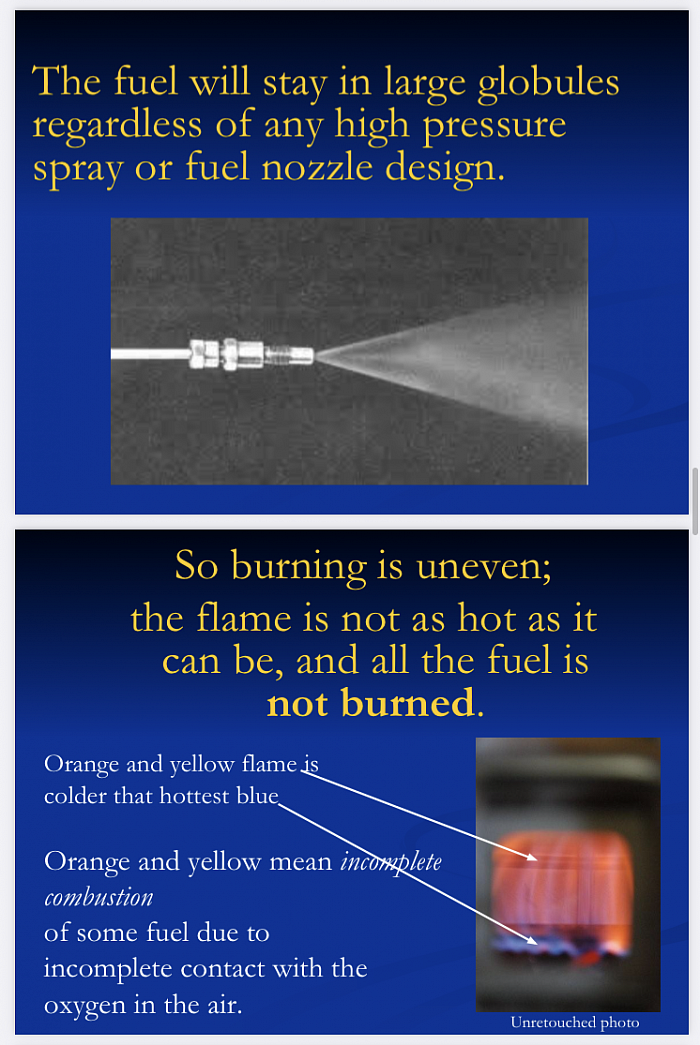
Only the molecules on the outer edges of this ball, where the oxygen in the air contacts the fuel molecules, actually burn.
As the molecules of fuel on the inside of this glob of molecules do not contact the oxygen in the air, they either:
-Do not burn at all, putting raw fuel into the environment as a “pollutant”
-Or burn incompletely creating, raw carbon, CO2, and other nasty chemicals we call pollution.
Contact Us:
Corporate Offices
Joshua R&D Technologies, LLC
5550 Granite Parkway, Suite 270,
Plano, Texas 75024 USA